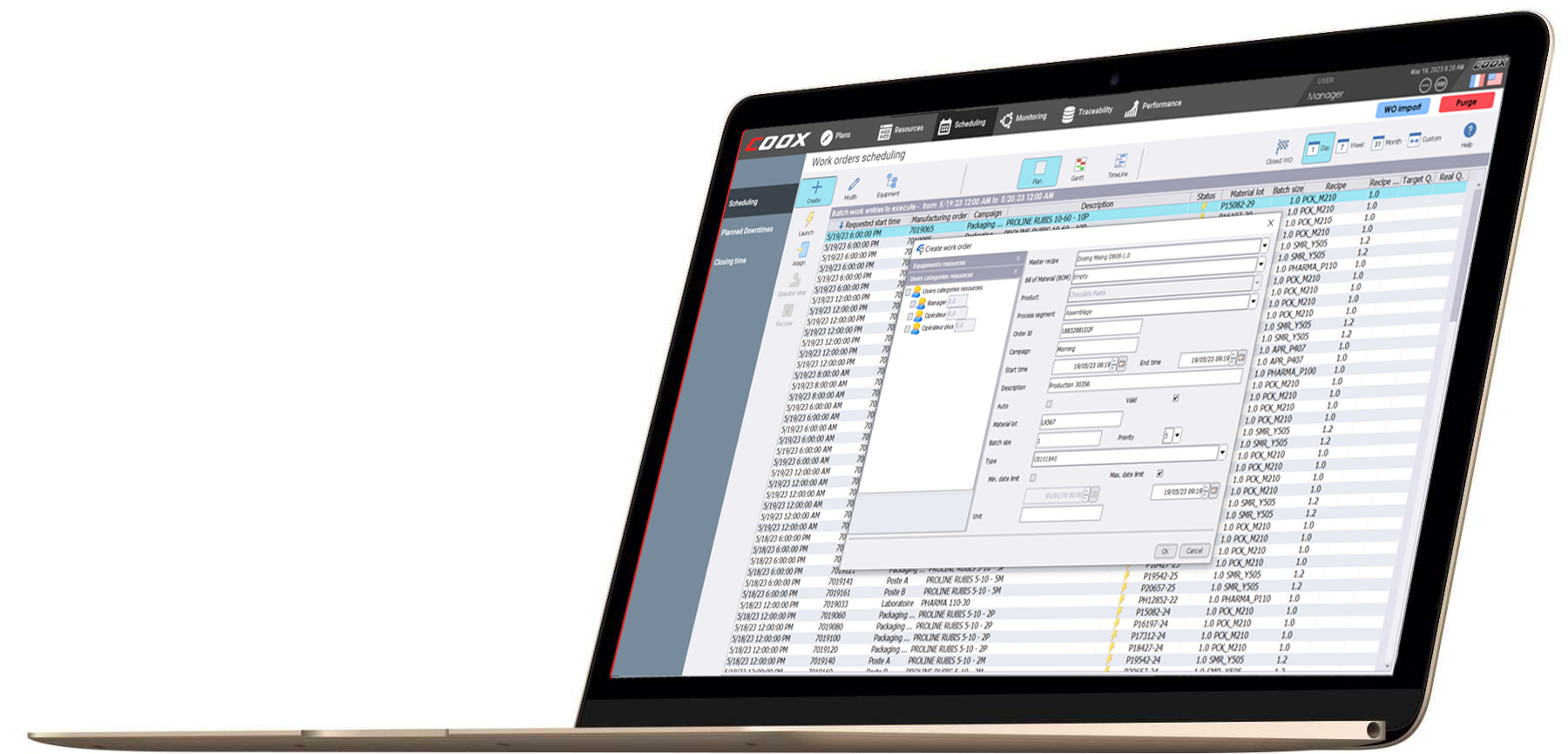
Production orders will allow to sequence the manufacture according to the orders to be delivered.
Import from ERP
Production orders are most often objects of exchange between ERP and production monitoring. (Manufacturing Execution System). Production orders are usually prepared in the ERP from orders and transferred to production monitoring (MES), but some industries favor the direct creation of production orders in the MES. Both modes are possible. In all cases, the direct creation of the production orders in the MES makes it possible to have an emergency mode in case of maintenance of the ERP. The import operation is often carried out periodically in the form of specific or standardzed files (B2MML format of the ISA-95 for example).
Launching and monitoring production orders
Production orders can be started manually or automatically (scheduled date and time). The detailed planning of production orders can be consulted at any time, as well as their progress (stages of manufacture). If there is a problem, or if a particular non-scheduled operation is to be performed, the production order can be suspended and then resumed. In all cases, this information is traced through the monitoring of the procedures and the process. The production orders can be split into several tasks and several production orders can be executed simultaneously. The allocation of the resources required for each production order is then automatically managed by the system.

CORRESPONDING MODULES OF THE COOX® RANGE
The production orders management functions are provided by the Manufacturing option of MESbox SCADA module and more fully by MESbox PMT module (Production Management & Traceability) of COOX range.
• Runtime software for batch and discreet processes
• Conformity ISA-88/ISA-95
• Graphic modeling of the process without data-processing knowledge
• Ergonomics of control customizable
• Detailed planning of the work orders
• Traceability of the set points and actual value, batch file
• Automatic quantitative adjustment
• Visualization and piloting since any point of the intranet
• Direct link with the traceability of product
• FDA requirements 21 CFR 11


